 Dissolving sugar in water increases the energy of the system as the randomness of the system increases and hence the entropy also increases.Ī campfire is also an example of entropy. A solid is actually in a much-ordered state, on dissolving, goes into a more disordered one. We shall never see the smoke being settled and concentrated at a place on its own.ĭissolving also leads to increased entropy. This is an example of the entropy increasing. It expands on its own as the molecules gain randomness and disorders. Osmosis − When we light an incense stick in a closed calm room, the smoke produced always tends to spread out. A few examples from day-to-day life can be listed as follows − In daily life, we see evidence that the universe always tends toward the way where there is an increase in the entropy, i.e. Various instances of entropy changing can be seen around us. 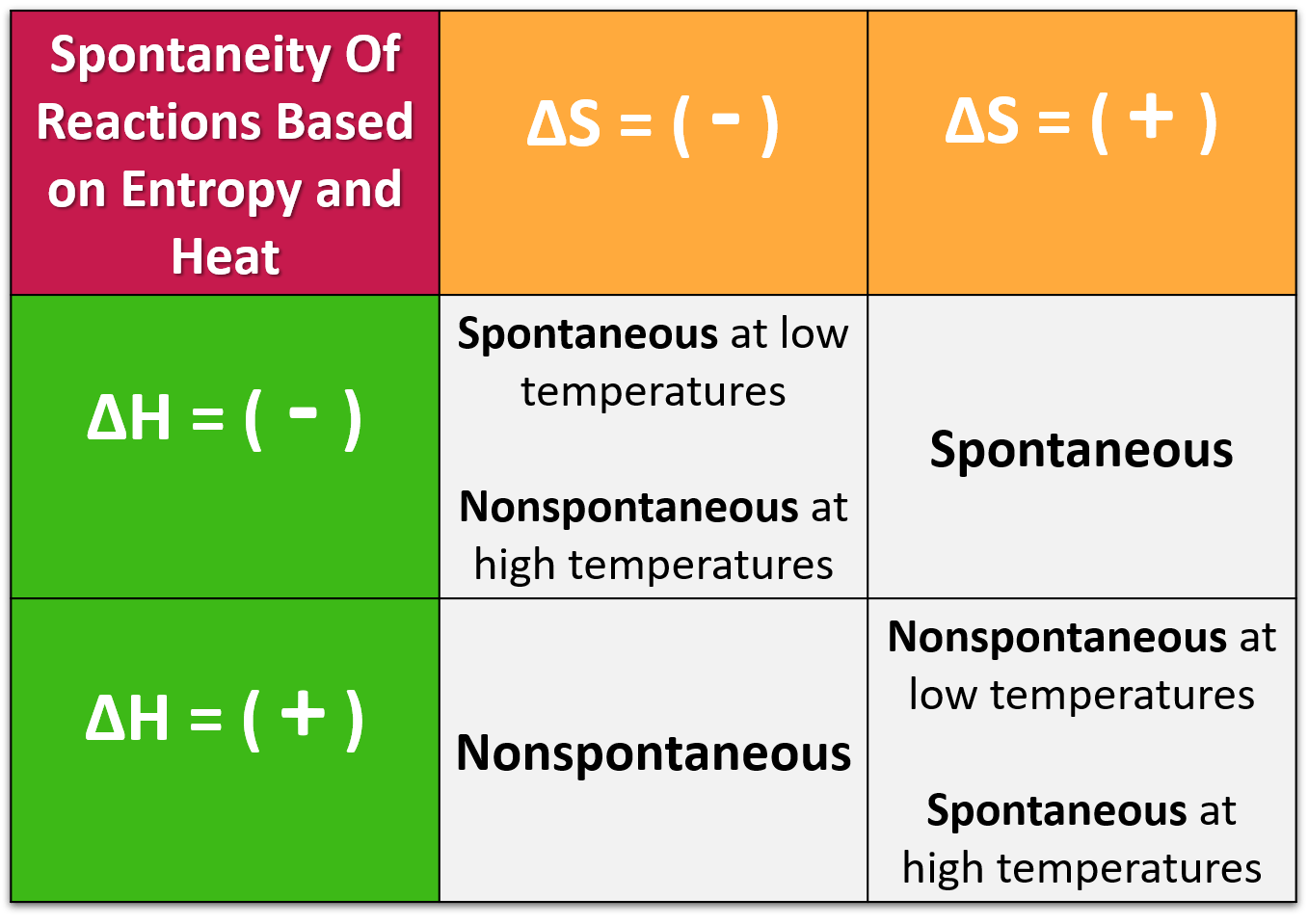
This implies that the entropy of the universe is always increasing. 
The entropy of the system, plus the surrounding, always increases. Hence the solids usually have less entropy than fluids. If the randomness of the molecules is less, the change in entropy will also be less. Later, Clausius, in his statement of the Second Law of Thermodynamics, it was defined it as the ratio of the very small change in the heat to the instantaneous temperature for a reversible process. Initially, the concept was coined with the name ‘heat potential. In this way, we may say that entropy is a function of the heat quantity. And hence, the part of heat which may be converted into work will not be much. The more disordered and random the molecules are, the more will be the entropy. Any kind of matter or medium- fluid or solid, is made up of molecules. What is Entropy?Įntropy is a measurable physical property that represents the degree of disorder of a system. In very simple words, the entropy is the measure of randomness on the other hand, the enthalpy represents the total heat of the system. In this article, the basic definitions of entropy and enthalpy, as well as the differences will be discussed in an ordered way. To understand it well, the concept of two terms- entropy and enthalpy has to be deeply dug in. Thermodynamics is a study that establishes a relationship between heat and work. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
